RECHERCHE
boerse.de
Europas erstes Finanzportal
Aktien-Ausblick
kostenlos, schon >100.000 Leser
boerse.de-Aktienbrief
kostenlos + unverbindlich
Börsendienste
kostenlos + unverbindlich kennenlernen
Leitfaden für Ihr Vermögen
einzigartiges Börsenwissen kostenlos
boerse.de-Investoren-Club
kostenlos Mitglied werden
INVESTMENT
boerse.de-Gold (TMG0LD)
Responsible Gold!
boerse.de-Fonds - ohne Agio!
boerse.de-Aktienfonds
boerse.de-Weltfonds
boerse.de-Technologiefonds
boerse.de-Dividendenfonds
boerse.de-Indizes
BCDI -
Das Original!
BCDI
USA
BCDI Deutschland
Einzelkontenverwaltungen
ab 500.000 Euro
myChampionsPREMIUM
boerse.de-Depotmanagement
boerse.de-Stiftungs-Strategien
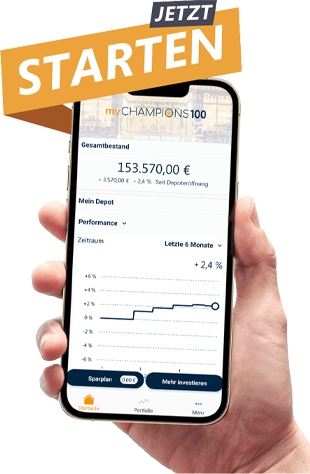
myChampions100
ab 100.000 Euro
myChampions100GOLD
NEU
myChampions100BITCOIN
NEU
myChampions100GOLD-BITCOIN
NEU
ÜBER UNS
RECHERCHE
Aktien-Ausblick
boerse.de-Aktienbrief
Börsendienste
Leitfaden für Ihr Vermögen
boerse.de-Investoren-Club
INVESTMENT
boerse.de-Fonds
boerse.de-Aktienfonds
boerse.de-Weltfonds
boerse.de-Technologiefonds
boerse.de-Dividendenfonds
boerse.de-Indizes
myChampionsPREMIUM
boerse.de-Depotmanagement
boerse.de-Stiftungs-Strategien
myChampions100
myChampions100GOLD NEU
myChampions100BITCOIN NEU
myChampions100GOLD-BITCOIN NEU
ÜBER UNS
Adhocs
Aktien-News
Aktiensplits
Banken-Prognosen
Dividenden
Insider Trades
IPOs
Unternehmenstermine
Analysen & Tools
Aktien-Chartanalysen
Chart-Tool
Beliebteste Aktien
Kursmarken-Suche
Performance-Checks
Aktien-Ausblick
boerse.de-Aktienbrief
Börsendienste
Experten
Leitfaden
Investoren-Club
Verdoppeln ist das Mindeste!
Börsenwohlstand für Alle!
Investments & Strategien
BCDI
boerse.de-Fonds
boerse.de Vermögensverwaltung
myChampions100
myChampionsPREMIUM
Erfolgreiche Vermögensverwaltung
Depot eröffnen
Aktienkurse (Übersicht)
Realtimekurse (Übersicht)
Deutsche Indizes
Dax 40 Realtimekurse
MDax Realtimekurse
TecDax Realtimekurse
SDax Realtimekurse
Internationale Indizes
Dow Jones Realtimekurse
Nasdaq Realtimekurse
S&P 500 Realtimekurse
MSCI World Realtimekurse
Euro Stoxx 50 Realtimekurse
BCI Realtimekurse
boerse.de-Aktienfonds (thesaurierend)
boerse.de-Aktienfonds (ausschüttend)
boerse.de-Weltfonds (thesaurierend)
boerse.de-Weltfonds (ausschüttend)
boerse.de-Technologiefonds (thesaurierend)
boerse.de-Technologiefonds (ausschüttend)
boerse.de-Dividendenfonds (thesaurierend)
boerse.de-Dividendenfonds (ausschüttend)
BCDI-Indizes
BCDI Realtimekurse
BCDI Deutschland Realtimekurse
BCDI USA Realtimekurse
Aktien-Kategorien
Technologie-Aktien
Deutsche Aktien
US-Aktien
Börsentermine
Börsenlexikon
Börsengeschichte
Börsengurus
Börsenhandelszeiten
Börsenvision
Champions-Aktien
Finanzrechner
Investment-Philosophie
Leitfaden für Ihr Vermögen
Performance-Analyse
Staatsverschuldung
Technische Indikatoren









 Suche
Suche

 Teilen
Teilen
 Link kopieren
Link kopieren
 boerse.de-Newsletter
boerse.de-Newsletter